NIH3T3 Origin
NIH 3T3 mouse embryonic fibroblast cells were initiated from a cell line isolated in 1962 at the New York University School of Medicine Department of Pathology. 3T3 refers to the cell transfer and inoculation protocol for the line, and means “3-day transfer, inoculum 3 x 105 cells.” Using this protocol, the immortal cell line begins to thrive and stabilize in cell culture after about 20-30 generations of in vitro growth. George Todaro and Howard Green, the scientists who first cultured this cell line, obtained the cells from desegregated NIH Swiss mouse embryo fibroblasts. The cell line has since become a standard fibroblast cell line.
NIH3T3 Characteristics
As one of the most commonly utilized cell lines, the NIH3T3 cell line has been incorporated in studies for a range of mechanistic and cell based assays, including protein functional analysis. The morphology of the cells are adherent, fibroblastic and are considered to be among the relatively easy to grow cell lines.
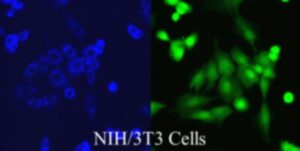
Copyright picture from altogen.com Reproduced with permission from Altogen Biosystems.
The original cells are extremely contact inhibited, although the cell line is no longer inhibited. NIH 3T3 cells are receptive to transformation with polyomavirus and SV40. Also, 3T3 cells are sensitive to sarcoma virus focus formation, leukemia virus and are inhibited by temazepam and other benzodiazepines.
The NIH 3T3 cells doubling time is 20-26 hours. Cell lines have a set of conditions for which they grow best. This includes a specific serum to provide additional growth factors, growth media, antibiotics to prevent bacterial growth and CO2 at a percentage for which the cells thrive (i.e. 5% CO2). Some cell lines require supplements such as non-essential amino acids (NEAA) or glutamine. Mammalian cells are grown in incubators set to 37°C.
Expression
Lysophosphatidylcholine (lyso-PC) induces AP-1 activity and c-jun N-terminal kinase activity (JNK1) by a protein kinase C-independent pathway.
NIH3T3 Cytogenetics
3T3 mouse cells are hypertriploid. The modal chromosome number is 68, which occurs in 30% of cells. Higher ploidies occur at a much lower rate of 2.4%.
NIH 3T3 Cell Migration
Cell interactions with a surface influences behaviors such as proliferation, migration and differentiation. In order to study the behavior of a cell in relation to cell migration, the scrape or wound healing assay can be incorporated. Although there are many variations of the assay, the classic scrape assay is performed by making a scratch or wound in the monolayer of cultured cells. Using photographic data, cell migration into the area of the wound can be quantified using images captured during the experiment.
The scrape assay is an older technology with many flaws incorporated during the formation of the wound (i.e. inconsistent pressure applied to well bottom, cell confluency at wound site, etc). To combat these inconsistencies, there are now chemical and automated ways to create the wound for migration studies. Also, there are cartridge style assays in which cells exhibit migration through a membrane in pursuit of fresh culture medium.
Links
Commercially available NIH/3T3 transfection reagent is available from Altogen Biosystems
siRNA Delivery – In Vivo Transfection Kits
CRO Pre-clinical Research Services: Xenograft animal models
Generation of Stably Expressing Cell Lines in 28 Days
Encapsulation of Protein, RNA, mRNA, and DNA Molecules into Liposomes
NIH 3T3 (Mouse embryonic fibroblast cell line) Whole Cell Lysate
Research Articles
- Cytogenic characterization of the cell line: In this study, scientists studied the genome of NIH 3T3 cells. They characterized the gains and losses of copy numbers in NIH 3T3, and found that it had only gained four derivative chromosomes since its initial cytogenic study in 1989. Efforts to align NIH 3T3’s genome imbalances with homologous regions in humans indicated that imbalances in NIH 3T3 were similar to those in human solid cancers of the ectoderm.
LINK
• Comparison of NIH 3T3 cells to primary embryo cells: In an early study, the NIH 3T3 cells were compared to both other 3T3 cells and primary embryo cells. It was found that sensitivity to sarcoma virus formation and leukemia virus growth were comparable between NIH 3T3 cells and the other cells studied.
LINK
• Identification of Canine Distemper Virus (CDV) receptors for uptake: To determine what receptors CDV utilizes for uptake into susceptible cells, a monoclonal antibody was identified that binds to the cell surface and inhibits CDV infection. Once it was identified that CD9 was the receptor bound by this antibody, a plasmid was transfected into NIH 3T3 cells to express CD9. These cells were subsequently determined to be more permissive to CDV, however immunoprecipitation and virus-overlay protein binding assays were unable to determine if CDV is directly binding to the CD9 surface receptor.
LINK
• Mesothelioma glycoprotein antigen production: In this study, a cDNA fragment that encodes a glycoprotein common to mesothelial cells, mesothelioma and ovarian cancer cells was identified. The cDNA was then transfected into NIH 3T3 cells, which produced the glycoprotein on their surface. The glycoprotein was subsequently named mesothelin and may play a role in cellular adhesion.
LINK
• Reversible immortalization: NIH 3T3 cells were used as a control in a study of reversible immortalization of mammalian cells using an oncogene. Scientists were able to determine that the oncogene provided immortalization, and then, once excised, cells reverted to their pre-immortalization characteristics.
LINK
NIH3T3 Cells │ NIH3T3 Cell Culture │ NIH3T3 Transfection │ Get NIH 3T3 Transfection Reagent